| Location: Home >> News >> Research Progress | ||||
| Research Progress | ||||
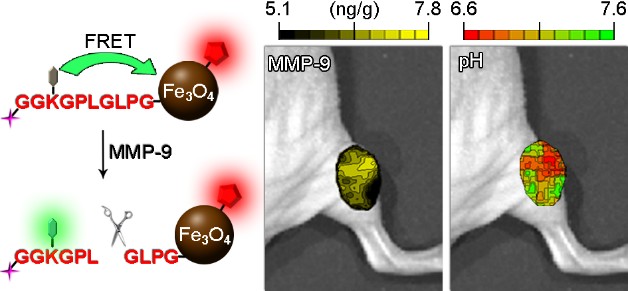
| Dual-Ratiometric Target-Triggered Fluorescent Probe for Simultaneous Quantitative Visualization of Tumor Microenvironment Protease Activity and pH in vivo
http://www.gaomingyuan.com Sunday, Dec 10, 2017 10:00 |
||||
 |
||||
The abnormal expression of tumor-associated proteases and lowered extracellular pH are important signatures strongly associated with cancer invasion, progression, and metastasis. However, their malignant effects were mainly identified using cell and tissue studies. To non-invasively visualize the heterogeneous distribution of these abnormal indicators in vivo and further disclose their collective behaviors, a protease-triggered dual-ratiometric fluorescent probe has successfully been constructed based on a FRET system comprised of Fe3O4 nanoparticles, pH-sensitive ratiometric fluorescent dye, NIR dye, MMP-9 specific peptide substrate linker, and folic acid as a tumor active targeting moiety. In addition to MRI compatibility enabled by Fe3O4 portion of the nanoprobe, this rational design allows these intravenously delivered nanoparticles to simultaneously map the protease activity of MMP-9 and microenvironmental pH of tumors. Systematic studies further demonstrate that the dual-ratiometric systems provide not only instant but also quantifiable information on the local protease activity of MMP-9 and pH in tumors. The current study also demonstrated that the overexpression of MMP-9 is well-correlated, in both time and location, with abnormal pH in vivo. The synergistic effects of these two characteristics largely govern the heterogeneous invasion of malignant tumors. Therefore, the key novelty of the current study lies in the quantitative and real-time detection of multiple tumor microenvironmental factors through non-invasive multimodality imaging. Thus, we believe that the general concept of the probe design demonstrated herein may represent a powerful tool for studying and characterizing abnormal tumor signatures in vivo. Tiancong Ma et al. |
||||